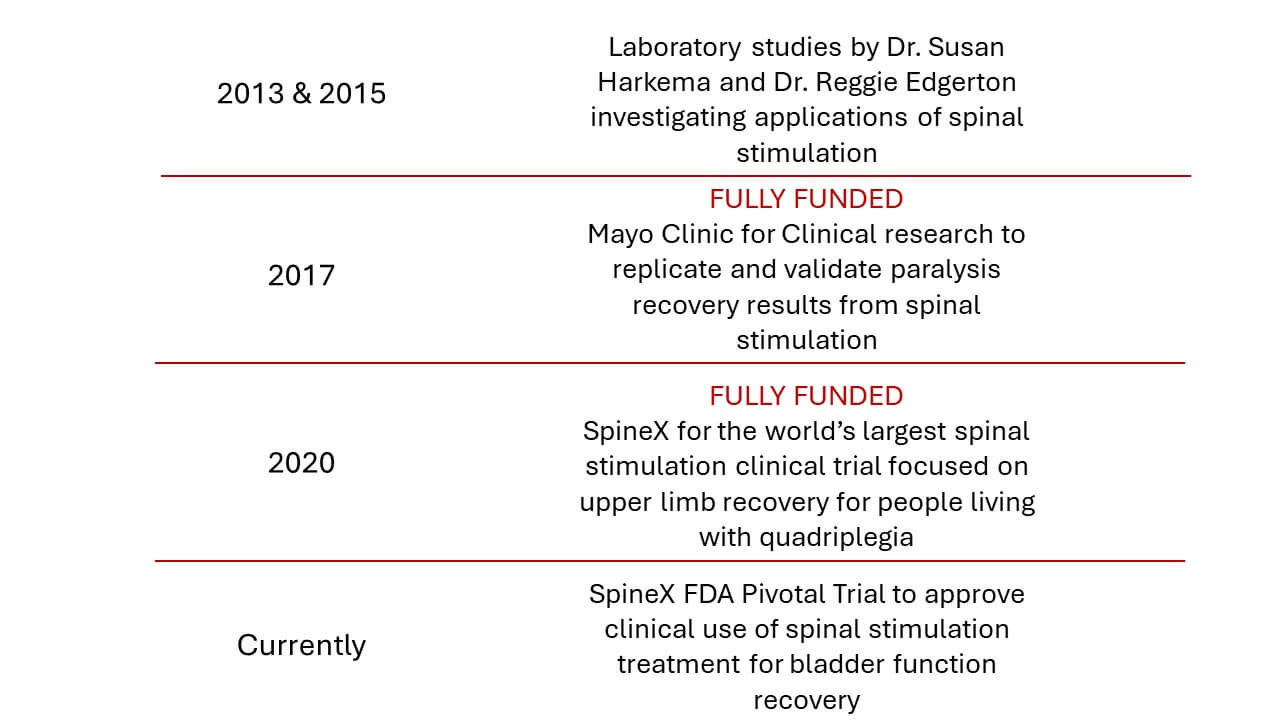
WE ARE FUNDING A GROUND-BREAKING CLINICAL TRIAL

The Jack Jablonski Foundation has partnered with top researchers to discover treatments that have proven to provide patients new functionality of their arms and legs and as a result, they’ve gained a renewed sense of independence and quality of life.
Our current clinical trials are exploring real world applications of transcutaneous spinal stimulation therapies to restore function using electricity to amplify nerve signals both above and below the point of an injury to the spinal cord. The therapy is administered using SCONE, an innovative device that received FDA Breakthrough Device Designation, meaning there is no similar competitive device on the market and the proposed therapy is significantly better than the current standard of care.
In the two clinical trials that the Jack Jablonski Foundation is funding, SCONE has demonstrated recovery of upper limb (hand and arm) function for people living with chronic quadriplegia and recovery of bladder function for people living with chronic paraplegia and other neurogenic conditions like stroke and MS.
Patient recovery data from these trials will be submitted to the FDA to seek approval for these new clinical treatments to become available to all Americans living with chronic paralysis and neurogenic bladder conditions.
SCROLL TO SEE OUR PROGRESS REPORT
PROGRESS REPORT
SpineX SCONE
Upper Limb Clinical Trial
Problem: Individuals living with quadriplegia (also known as tetraplegia) have lost the ability to control their arms, hands, trunk, legs, and pelvic organs. Impaired hand function severely limits the ability to perform simple tasks of everyday living resulting in a loss of independence.
Solution: Transcutaneous neuromodulation, a non-invasive electrical stimulation therapy, has demonstrated improved hand function in clinical trial participants.
Outcome: All 24 participants in our SpineX upper limb clinical trial responded positively to treatments using the SCONE device. Because all spinal cord injuries are unique, the extent of hand function recovery and improved core strength varied by participant and their level of injury. But every participant in the study regained abilities they were once told were permanently lost to paralysis. Every participant regained functions that allowed them to perform some simple activities of daily living again for the first time since being injured.
Study Description: FULLY FUNDED BY THE JACK JABLONSKI FOUNDATION
SpineX SCONE
Pivotal Trial for Neurogenic Bladder Treatment
Problem: Individuals living with paralysis resulting from a spinal cord injury have lost the ability to control all or part of their trunk, legs, and pelvic organs. This includes a loss of bladder control, a condition called neurogenic bladder. People living with neurogenic bladder experience a lost sensation of bladder fullness, low bladder capacity, and frequent urination cycles. They also live with a constant fear of uncontrolled urine leakage. This requires most individuals to rely for a lifetime on daily repetitive catheterizations to empty their bladder. This repetitive catheterization dramatically reduces independence for people living with paralysis and is often the cause of other serious medical consequences.
Solution: Transcutaneous neuromodulation using the SCONE device, a non-invasive electrical stimulation therapy, has demonstrated improved bladder control and function in clinical trial participants. The treatment has improved the quality of their lives.
Outcome: Our SCONE study is the first large-scale clinical trial testing a non-invasive spinal neuromodulation technology for the treatment of neurogenic bladder. The purpose is to study the effectiveness and safety of SCONE neuromodulation therapy for individuals with neurogenic lower urinary tract dysfunction due to a spinal cord injury as well as stroke and multiple sclerosis. So far, more than 80% of people who have used SCONE experienced improvements in neurogenic bladder symptoms. Completion of this study will move the SCONE device one step closer to our goal to gain FDA approval so that it can become available for use on everyone living with the loss of bladder control.
SpineX has successfully completed the enrollment of individuals to participate in the SCONE Continence study at a dozen independent clinical sites, including ten in America. This marks a significant milestone in the journey toward obtaining de Novo registration from the FDA.
The Jack Jablonski Foundation is helping to fund this groundbreaking pivotal trial that includes 90 participants.
To make a gift and help us get this done, donate today.
Spinal Stimulation Path to Clinical Treatment
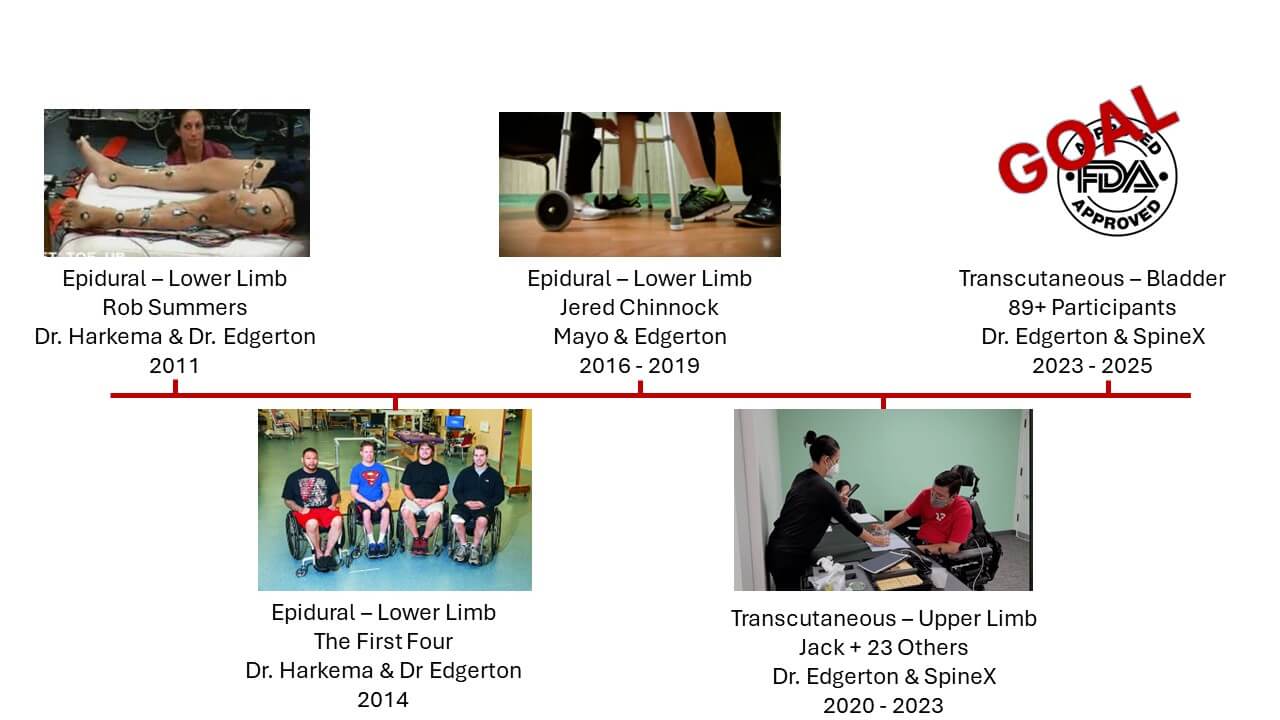
Our Research Grants